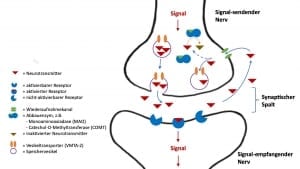
Stoffwechselwege von aktivierendem Neurotransmitter – ein einfaches Modell
In der der Cardiopraxis befassen wir uns zunehmend mit den Stoffwechselwegen der aktivierenden Neurotransmitter Dopamin, Noradrenalin, Serotonin und Adrenalin. Diese haben nicht nur einen Einfluss auf Ihr emotionales Befinden und Verhalten, sondern auch auf das Herz-Kreislaufsystem, wie z.B. den Blutdruck und Herzrhythmus.
Um die Wirkung von aktivierenden Neurotransmittern besser verstehen zu können, müssen wir uns mit den Prinzipien ihres Stoffwechsels befassen.
Neurotransmitter sind Botenstoffe zur Signalübertragung
Neurotransmitter sind Botenstoffe, die eine Nachricht vermitteln, entweder zwischen 2 Nerven oder zwischen einem Nerv und einem Endorgan, z.B. dem Herz. Kurzum, Neurotransmitter dienen der Kommunikation, vor allen Dingen im Gehirn, dem Hauptort der Informationsverarbeitung für Außeneinflüsse der Umwelt. Darüber hinaus sind sie z.T. auch über das vegetative Nervensystem Signalübermittler zwischen Gehirn und den übrigen Anteilen des Körpers.
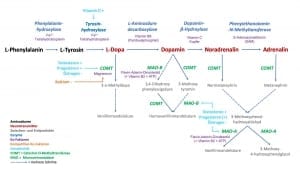
Meistens sind es mehrere Stoffwechselschritte für die Bildung von aktivierenden Neurotransmitter aus jeweils einer Aminosäure notwendig. So nehmen wir z.B. die Aminosäure L-Phenylalanin mit der Nahrung auf und aus ihr entsteht durch verschiedene Umbauprozesse z.B. der Neurotransmitter Dopamin. Da die aktivierenden Neurotransmitter nur eine Aminosäure beinhalten, nennen wir sie folglich auch Monoamine.
Stoffwechsel von Neurotransmittern – ein einfaches Modell
Betrachten wir den Stoffwechsel von aktivierenden Neurotransmittern, dann können wir grundsätzlich 4 Ebenen unterscheiden:
- Bildung
- Wirkung
- Speicherung
- Abbau
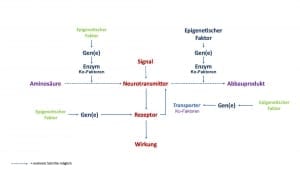
An diesen Prozessen sind verschiedene Faktoren beteiligt. Vereinfacht betrachtet sind es:
- Nährstoffe
- Gene
- epigenetische Faktoren
- Enzyme
- Enzym Ko-Faktoren
- Rezeptoren
Dabei müssen alle Prozesse und deren Faktoren im Gleichgewicht sein, damit Sie gesund und leistungsfähig sind.
Enzyme und Ko-Faktoren
Enzyme sorgen dafür, dass Neurotransmitter meist über mehrere Stoffwechselschritte gebildet werden. Diese Enzyme sind großmolekulare Eiweiße, die die Bildung als Katalysatoren erheblich beschleunigen, ohne dass sie sich selber dabei strukturell oder funktionell verändern. Hat ein Enzym einen Stoffwechselschritt durchgeführt, dann steht es für eine erneute Stoffwechselleistung wieder zur Verfügung, solange die richtigen Ko-Faktoren vorhanden sind.
Enzyme benötigen in der Regel einen oder mehrere Ko-Faktoren, um richtig zu funktionieren. Diese Ko-Faktoren sind zwar für das einzelne Enzym spezifisch, können aber bei verschiedenen Enzymen ganz unterschiedlich sein. Sie kommen häufig aus der Gruppe der sog. Mikronährstoffe; die bekanntesten sind Vitamine und Mineralien, so z.B. Vitamin B6 oder Magnesium.
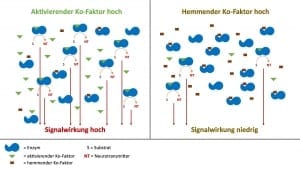
Für jedes Enzym gibt es einen oder mehrere optimale Ko-Faktoren. Oder anders gesagt, für jedes Schloss gibt es den passenden Schlüssel. Nur zusammen mit dem optimalen Ko-Faktor kann das einzelne Enzym den spezifischen Stoffwechselschritt mit maximaler Geschwindigkeit durchführen.
Nun gibt es auch nicht-optimale Ko-Faktoren, die eine Stoffwechselschritt verlangsamen oder sogar blockieren können. Optimale und nicht-optimale Ko-Faktoren konkurrieren um die Bindungsstelle am Enzym. So kann es z.B. sein, dass der optimale Ko-Faktor Magnesium durch den nicht-optimalen Ko-Faktor Kalzium verdrängt wird.
Enzyme – Ko-Faktoren als regulierender Faktor
In einer Zelle kommt derselbe Enzymtyp mehrmals vor, da ein einzelnes Enzym die notwendige Syntheseleistung alleine nicht erbringen könnte. Für die Geschwindigkeit eines Stoffwechselschritts ist neben der Substratkonzentration und der Temperatur auch das Gleichgewicht zwischen folgenden Zuständen von Enzymen wichtig:
- nicht besetzte Enzyme ohne Ko-Faktor
- Enzyme mit optimalem Ko-Faktor
- Enzyme mit nicht-optimalem Ko-Faktor
So konkurrieren optimale und nicht-optimale Ko-Faktoren um die Bindungsstelle an mehreren Enzymen eines Typs. Je nachdem wie diese Ko-Faktoren, optimale und nicht-optimale, in einem quantitativen Gleichgewicht zu einander stehen, läuft der Stoffwechselweg aller Enzyme desselben Typs in Summe schneller oder langsamer. Anders formuliert, sind viele nicht-optimale Ko-Faktoren vorhanden, dann läuft der Stoffwechselschritt langsam, sind viele optimale Ko-Faktoren vorhanden, dann läuft er schneller.
Hieraus wird deutlich, dass Ko-Faktoren eine wichtige Regulierungsfunktion für Stoffwechselwege haben. Diese Zusammenhänge haben vor dem Hintergrund der unkontrollierten Zufuhr von freiverkäuflichen Nahrungsergänzungsmitteln eine erhebliche Bedeutung. Wird das Gleichgewicht, sei es entweder durch ein Übergewicht von nicht-optimalen aber auch von optimalen Ko-Faktoren eines Enzyms gestört, dann können negative gesundheitliche Folgen auftreten.
Wir kennen das aus der Praxis sehr gut z.B. von der übermäßigen Zufuhr von Vitamin B6 (Umwandlung von L-Dopa zu Dopamin durch L-Aminodecarboxylase). Nicht nur kann die Überbehandlung mit Vitamin B6 schwere neurologische Störungen zur Folge haben, sondern es tritt hierunter auch eine übersteigerte Stoffwechselaktivität auf, die z.T. vermittelt durch Neurotransmittern innere Unruhe, Gereiztheit, Schlafstörungen, Schwitzen und Kreislaufprobleme nach sich zieht.
Gene und epigenetische Faktoren steuern die Bildung von Enzymen
Enzyme werden als Eiweißstoffe selber durch ein einzelnes Gen bzw. mehrere Gene gebildet. Spezifische Gene liegen im Zellkern einer Zelle und sind für die Bildung von Enzymen gewissermaßen Schnittmustervorlagen.
Gene können in ihrer Funktion an- und abgeschaltet werden. Die Faktoren, die das bewirken sind unter anderem “epigenetische Faktoren“. Epigenetik bedeutet vereinfacht “beim Gen“. Ähnlich wie die Ko-Faktoren bei den Enzymen haben sie somit eine regulierende Funktion.
Die Epigenetik ist ein verhältnismäßige neues Wissenschaftsgebiet. Gerade in der Tumorforschung wird hier nach epigenetischen Einflussfaktoren sowohl für die Bildung als auch die Behandlung von Tumorerkrankungen intensiv geforscht.
Bei der Regulierung des Herz- Kreislaufsystem durch aktivierende Neurotransmitter spielen epigenetische Einflussgrößen ebenfalls eine wichtige Rolle. Nach unseren Erfahrungen in Verbindung mit systematischen wissenschaftlichen Erkenntnissen haben hier z.B. Vitamin D und die Geschlechtshormone Östrogen und Testosteron eine hervorzuhebende Bedeutung.
Vitamin D induziert die Bildung des Enzyms Tyrosin-Hydroxylase (Umwandlung von Tyrosin in L-Dopa). Testosteron induziert genetisch die vermehrte Bildung der Catechol-O-Methytransferase (COMT) und Monoaminooxidase (MAO), was wiederum einem beschleunigten Abbau aktivierenden Neurotransmitter verbunden ist. Östrogen hingegen verlangsamt über eine verringerte Synthese von COMT und MAO den Abbau von aktivierenden Neurotransmittern.
Sie können sich sicherlich vorstellen, dass diese Zusammenhänge nicht nur für Ihr emotional gesteuertes Verhalten, sondern auch die Regulierung des Herz-Kreislaufsystems eine wirkmächtige Bedeutung haben.
Signalstärke von Neurotransmittern
Sind die aktivierenden Neurotransmitter einmal gebildet, dann werden sie durch ein Signal im Nerven aus der Senderzelle in den Zellzwischenraum, den sog. synaptischen Spalt freigesetzt, um das Signal an eine Empfängerzelle zu übertragen. An der Empfängerzelle sitzen in der Zellmembran Rezeptoren, an welche die Neurotransmitter andocken. Über die Rezeptoren werden dann weitere Signalwege innerhalb der Zielzelle aktiviert.
Für die Signalstärke, und ob überhaupt eine Signalübertragung zustande kommt sind mehrere Faktoren wichtig:
- Signal-sendende Nerven
- Neurotransmitter
- Signal-empfangende Nerven
- Spezifische Rezeptoren für Neurotransmitter
Bei einem Signal durch einen Signal-sendenden Nerv erfolgt immer die Freisetzung mehrerer Neurotransmitter. Für die Entstehung eines Signals spielt zunächst einmal das Vorhandensein von Signal-empfangenden Nerven eine Rolle: ohne Empfänger keine Signalweiterleitung.
Da in der Regel ganze Nervenbündel für die Bildung bzw. Weiterleitung einer bestimmten Signalqualität verantwortlich sind, spielt das zahlenmäßige Verhältnis von sendenden und empfangenden Nerven auch eine Rolle, wobei auch mehrere Sender an einem Empfänger andocken können
Weiterhin hat auch das quantitative Verhältnis von Neurotransmittern zu ihren spezifischen Rezeptoren eine Bedeutung. So kann z.B. trotz zahlreicher Neurotransmitter im synaptischen Spalt ein Signal nicht übertragen werden, wenn auf der Signal-empfangenden Seite wenige oder gar keine Rezeptoren vorhanden sind.
Rezeptoren für Neurotransmitter
Wir ein aktivierender Neurotransmitter durch ein Signal aus dem Signal-sendenden Nerv freigesetzt, dann befindet er sich zunächst im Zwischenraum zwischen 2 Nerven, dem sog. synaptischen Spalt. Er dockt dann, gemäß dem Schlüssel-Schloss-Prinzip an spezifische Rezeptoren auf der Signal-empfangenden Zelle an. Der Rezeptor löst dann in der Signal-empfangenden Zelle eine intrazelluläre Signal-Kette aus und das Signal wird dann im Nerv weitergeleitet.
Rezeptoren werden selber durch Gene gebildet und werden selber durch spezifische epigenetische Faktoren und zum Teil über Rezeptor-spezifisch Ko-faktoren reguliert. Weitere Regulierungsmöglichkeiten sind die Ansprechbarkeit von Rezeptoren und die die Rezeptorblockade.
Wenn ein Rezeptor vermittelt durch einen Neurotransmitter ein Signal in die Signal-empfangenden Zelle übertragen hat, dann ist er danach für eine bestimmte Zeit inaktiv, d.h. er steht für eine erneute Signalübertragung nicht zur Verfügung. War das gesendete Signal aller Signal-sendenden Nerven quantitativ sehr stark, dann kann im Sinne einer Erschöpfung der ganze Signalweg für eine bestimmte Zeit blockiert sein. Folglich bleiben weitere Signale durch den Signal-sendenden Nerv wirkungslos.
Blockade von Neurotransmitterrezeptoren als therapeutisches Prinzip
Neurotransmitter und Rezeptor sind im Sinne des Schlüssel-Schloss-Prinzips spezifisch füreinander optimal passend. Ähnlich wie bei den Ko-Faktoren der Enzyme kann aber eine nicht-optimal passende Substanz den Rezeptor für den optimal passenden Neurotransmitter und damit die Wirkung desselben blockieren. Diese Blockade kann kompetitiv sein, d.h. die Signalübertragung wird durch das quantitative Verhältnis von optimal-wirkenden Neurotransmittern zu blockierenden Substanzen bestimmt. Allerdings gibt es auch eine nicht-kompetitive Hemmung, bei der der Rezeptor irreversibel blockiert ist, was in gewisser Weise einer Vergiftung entspricht.
In der Herz-Kreislaufmedizin wird das Prinzip der kompetitiven Rezeptorblockade, z.B. bei der Therapie mit Beta-Rezeptoren Blockern zur Behandlung von Bluthochdruck und Herzrhythmusstörungen regelmäßig eingesetzt. Wenn die Beta-Rezeptoren am Herz besetzt sind, dann können Noradrenalin und Adrenalin am Herzen nicht ihre volle Wirkung mit Steigerung von Herzfrequenz und Pumpkraft entfalten.
Wir können die kompetitive Hemmung durch den Beta-Rezeptoren Blocker übrigens sehr schön in der Stressechokardiografie beobachten. Menschen unter Beta-Blockertherapie weisen hier zunächst in Ruhe eine träge Pumpleistung auf. Mit zunehmender Belastung, und damit adrenerger Aktivierung gewinnen Noradrenalin und Adrenalin an den Herzmuskelzellen die Oberhand und die Pumpkraft steigt deutlich an; manchmal hat man als Untersucher den Eindruck, als sei ein Schalter umgelegt.
Wiederaufnahme von Neurotransmittern
Hat ein Neurotransmitter seine Funktion am Rezeptor des Signal-empfangenden Nerv erfüllt, dann löst er sich wieder, so dass er sich zunächst frei im Zwischenraum zwischen Signal-empfangenden und Signal-sendendem Nerven, dem sog. synaptischen Spalt befindet. In Einzelfällen finden sich an der Außenseite der Zellmembranen, der äußeren Zellgrenze hier Enzyme, die den Abbau des Neurotransmitters bewirken.
In den meisten Fällen werden die freien Neurotransmitter allerdings über einen Wiederaufnahmekanal erneut in den Signal-sendenden Nerv aufgenommen. Bei Wiederaufnahmekanälen handelt es sich um Eiweißstrukturen, die ebenfalls auf der Grundlage von Genen gebildet werden. Somit kann durch genetische Varianten die Wiederaufnahme von Neurotransmittern von Mensch-zu-Mensch unterschiedlich sein: beim einen ist die Wiederaufnahme schneller, beim anderen langsamer.
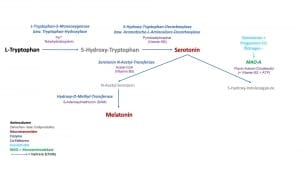
Die Funktion von Wiederaufnahmekanälen wird bei den aktivierenden Neurotransmittern auch therapeutisch genutzt. So verwenden wir bei der Behandlung von Depression die sog. Serotonin-bzw. die Noradrenalin-Wiederaufnahme-Hemmer. Durch die Blockade der Wiederaufnahme wird die Konzentration des Neurotransmitters im synaptischen Spalt erhöht und die Wirkung am Signal-empfangenden Nerv gesteigert.
Werden die Neurotransmitter über den Wiederaufnahme-Kanal wieder in den Signal-sendenden Nerv aufgenommen, dann werden sie entweder gespeichert oder abgebaut.
Speicherung von Neurotransmittern
In der Regel speichert der Signal-sendenden Nerv die Neurotransmitter nach ihrer Wiederaufnahme. Dieses geschieht in kleinen Speicherbläschen, sog. Speichervesikeln. Um in ein Speichervesikel zu gelangen, muss der Neurotransmitter erneut einen Transporter-Kanal, der ebenfalls aus Proteinen besteht, passieren. Der Transporter, in der Regel der Vesikuläre Monoamintransporter 2 (VMAT2) ist für alle aktivierenden Neurotransmitter (Dopamin, Noradrenalin, Adrenalin und Serotonin) gleich.
In den Vesikeln sind die Neurotransmitter vor den abbauenden Enzymen, die innerhalb der Zelle, aber außerhalb der Vesikel liegen, geschützt. Wird der Signal-sendende Nerv erneut aktiviert, dann stehen die Neurotransmitter in den Vesikeln für eine erneute Signalübermittlung an den Signal-empfangenden Nerv zur Verfügung.
Wir nutzen die Funktion eines Vesikel-Transporters auch therapeutisch. Der Blockade des Transporters VMAT2, z.B. durch Reserpin kann bei zu starker neurovegetativer Aktivierung z.B. zur Behandlung des Bluthochdrucks oder bei innerer Unruhe und Angstzuständen therapeutisch eingesetzt werden. Kurzum, Reserpin blockiert die Aufnahme von aktivierenden Neurotransmittern in die schützenden Vesikel. Folglich bauen dann Enzyme die Neurotransmitter außerhalb der Vesikel vermehrt ab.
Abbau von Neurotransmittern
Der Abbau von aktivierenden Neurotransmittern erfolgt überwiegend innerhalb des Signal-sendenden Nervs. Ebenso wie bei der Bildung von Neurotransmittern spielen bei ihrem Abbau Enzyme eine entscheidende Rolle. Dabei sind wieder die Aktivität, Gene und Ko-faktoren von regulierender Bedeutung.
Von hervorgehobener Bedeutung sind hier die Enzyme Catechol-O-Methytransferase (COMT) und die Monoaminooxidasen (MAO). Für COMT und MAO kennen wir genetische Varianten. So gibt es bei Menschen alleine schon genetisch bedingt eine hohe, mittlere und niedrige Abbaurate von aktivierenden Neurotransmittern durch COMT bzw. MAO. Ist die Abbaurate z.B. niedrig, dann “stauen“ sich die Neurotransmitter vor dem Enzym und es liegt bei diesem Menschen eher ein gesteigertes Aktivitätsniveau vor. Ist die die Abbaurate hoch, dann ist das Aktivitätsniveau eher niedrig.
In Bezug auf das menschliche Verhalten bedeutet dieses eine höhere bzw. niedrigere Irritabilität und erklärt so auch zum Teil das unterschiedliche Temperament zwischen einzelnen Menschen. Gerade die genetischen Varianten von COMT und MAO werden in der Verhaltensforschung ausgiebig untersucht. So kann z.B. eine niedrige Aktivität von COMT mit einem “Stau“ von Dopamin und Noradrenalin innere Unruhe und eine Neigung zu Panikattacken mit erklären. Eine erhöhte Aktivität von MAO und damit verbunden die gesteigerte Abbaurate des Neurotransmitters Serotonin kann eine Prädisposition für Depressionen bedeuten.
Aktivierende Neurotransmitter – genetische Zusammenhänge gezielt therapeutisch nutzen
Die Kenntnis der genetischen Unterschiede beim Auf- und Abbau von aktivierenden Neurotransmittern ist eine sehr gute Option zur besseren Individualisierung von Therapiemaßnahmen in der Herz-Kreislaufmedizin.
Gerade bei der Behandlung von Herzrhythmusstörungen und Bluthochdruck, die ja nicht nur von Störungen des Organs Herz bzw. der Blutgefäße selber abhängig sind, hat die Kenntnis der individuellen genetischen Voraussetzung für die Wirkung von aktivierenden Neurotransmittern eine zunehmende therapeutische Bedeutung. Nicht nur werden so Verhaltensmuster eher erklärbar, sondern wir können gezielter Empfehlungen bei der medikamentösen Therapie, dem Einsatz von Nahrungsergänzungsmittel und zu den Wirkungen von Hormonpräparaten, seien es Östrogen, Progesteron oder Testosteron geben. Diese haben wiederum eine Wirkung auf Herz und Kreislauf.
Weiter zum MediaRezept „Neurotransmitter„
Literatur
Cardiopraxis – Kardiologen in Düsseldorf & Meerbusch